Raman Scattering
Molecular information can be obtained using different experimental techniques. Probably the most common ones are infrared spectroscopy and the Raman scattering spectroscopy.
The Raman Effect
Molecular vibrations can be excited either through the absorption of light or the inelastic scattering. Infrared spectroscopy is based on the direct absorption of light and the process is similar to light absorption in the UV-Vis range. The difference is that in the UV-Vis range light causes the so-called vibronic transitions that simultaneously involve changes in the vibrational and electronic energy levels. See simplified schematic diagram in Figure 1.
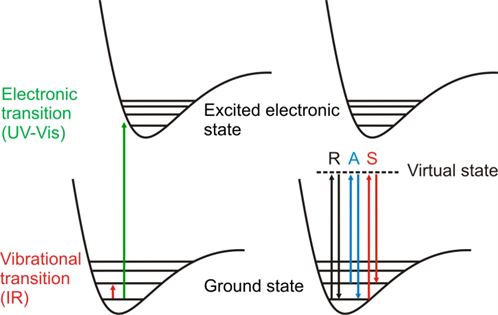
The Raman scattering phenomenon is different from IR spectroscopy. In a simplified manner it can be explained in terms of elastic (Rayleigh scattering) and inelastic (Raman scattering) collisions between incident light and target molecules. In the Rayleigh scattering, the incident light is transmitted through a target media without any change. While in the Raman scattering a small portion of light is scattered and the energy transfer between the two systems takes place. The Raman effect is a weak process that produces
scattering intensities 3-5 orders of magnitude smaller in comparison to the Rayleigh scattering. In the energy transfer process, the incident photon might lose energy by exciting higher molecular vibrational states. The photon then emerges with a lower frequency (longer wavelength) and the process is referred to as Stokes Raman scattering. The incident photon might also acquire the energy from the molecule and induce decay into lower molecular vibrational state. In this case the photon emerges with a higher frequency (shorter wavelength) and the process is known as anti-Stokes Raman scattering, see Figure 1 for schematic illustration.